AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Opposite of entropy12/3/2023  
There is a higher likelihood that the cubes will all be scattered on "floor level" than they will be stacked neatly on top of each other in any part of the box. After a few shakes, you open the box to see how the cubes are distributed. 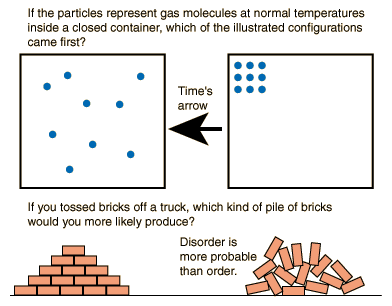
The bottom of the box is level and every now and then, someone shakes the box directly up and down - just causing a small disturbances. Think of a box filled with dozens of smaller cubes. Better copiers made more copies that could survive in a sea of other copies and a billion years later we arrived at this thread. As soon as that happened, you had an explosion of these molecules and suddenly the efficiency and ability to copy yourself became really important. In the briefest nutshell, at some point, some kind of molecule developed that could copy itself. At least in part anyways.įor further stimulating discussion on this, I would recommend Richard Dawkin's explanation of how life arose. So we're a fixed entropic cost that fuels itself and by the time we're done we've contributed more entropy back than we consumed. We can thus maintain our low entropic state because we're constantly breaking down other molecules (and thus adding their entropy back to the universe). We then pay back the loan in little entropic payments as we break down food for energy, with a little bit of interest. We take out a loan to keep our entropy low when we're created. The idea of negative entropy is that life fuels it's own relatively low entropy state by outsourcing its entropy elsewhere. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
